AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Understanding precipitate chemistry11/16/2023 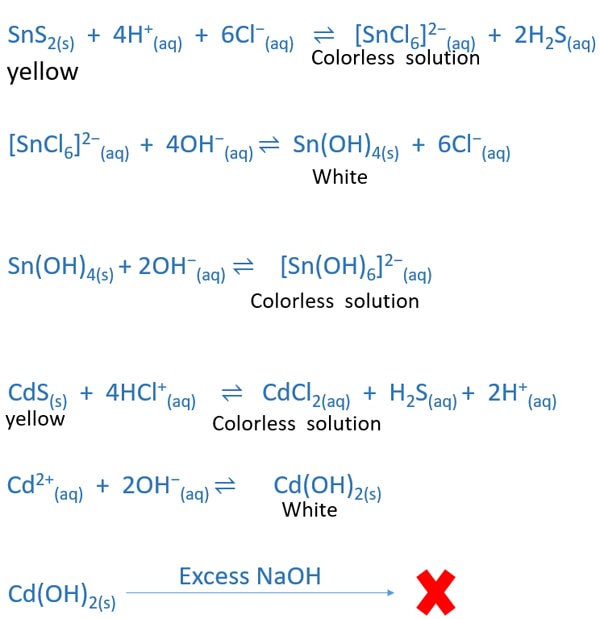
Activity 2: Identification of positive ions Now with the use of a plastic sheet, the procedure is rapid, giving the teacher time to generate student feedback on their observations, with very little waste generated. Using test tubes would generate a large amount of cleaning at the end. The fifth reaction is a ‘green’ reaction with chemicals available from the local shop. There will be no precipitate with the second one, which implies that sodium nitrate and potassium chloride are also soluble in water. Try reactions between the following salts: So, now students can complete a table of results and observations. It is really a saturated solution of copper carbonate in water.Įvery student knows sodium chloride is soluble in water. The reaction works because sodium carbonate and copper chloride are soluble in water, but copper carbonate is not very soluble, and this is the reason why a precipitate forms. The results allow students and teachers to discuss where the chemicals are coming from, and they provide more belief in the chemical equation: When clear solutions of copper(II) chloride (left) and sodium carbonate (right) are combined (centre), a blue precipitate of insoluble copper carbonate forms. Disposal: wipe the plastic surface with a paper towel and dispose of the towel in the waste.Stir the contents of the square with a freshly cut, pointed splint.With another pipette, move two drops of the sodium carbonate solution in the circle on the right into the square in the middle.With a pipette, move two drops of the copper chloride solution in the circle into the square in the middle. 
Add water with a pipette to fill the whole circle and stir with a freshly cut, pointed splint to dissolve the solid.Add copper chloride and sodium carbonate crystals to the boxes shown on the worksheet. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
